SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
Course Overview
Page content
| About this course - Chemistry 12 (SCH4U) | ||
|
.
. . |
||
| A word about Assignments | ||
|
This course is built with a number of formative tasks (check-up time activities) and summative assignments to develop your learning skills and to help you achieve high academic success on formal evaluations that decide your final grade. Formative tasks in each lesson demonstrate to you and your teacher how well you have understood the learning goals for the lesson. Completing formative tasks and summative assignments are critical to your learning. Students must complete all evaluation activities in each lesson in order to complete the unit tests and final examination. |
||
| Evaluation Framework | ||
|
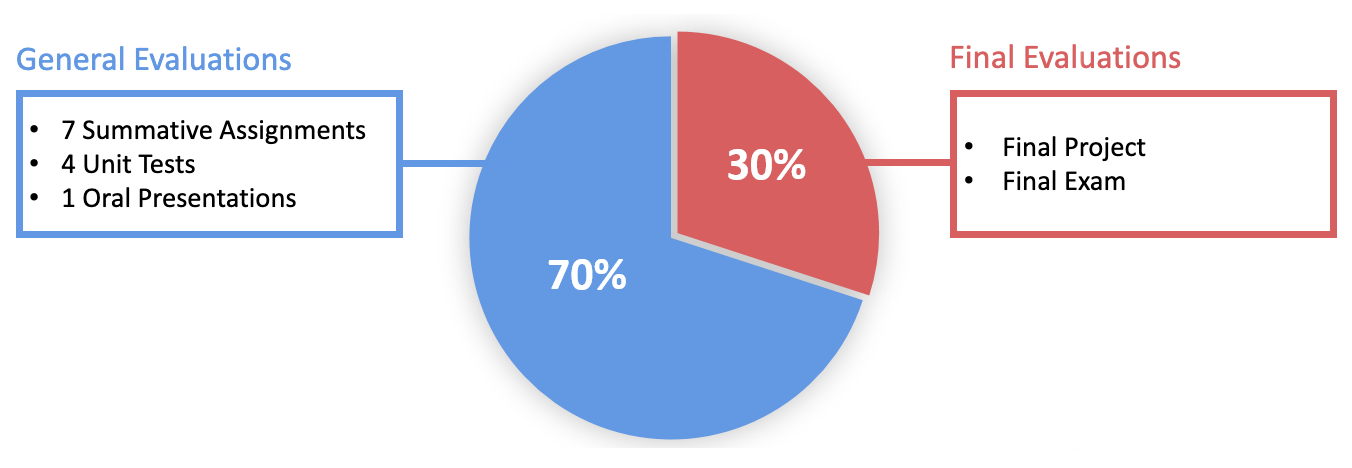
Grade Distribution The grade for this course is divided into two main parts: 70% for General Evaluations and 30% for Final Evaluations.
KICA Distribution This course follows a distribution of
Each evaluation in this course contains a KICA Achievement Chart that your teacher will use to grade your submission. The KICA Achievement Chart is a detailed evaluation guide with criteria and descriptors to grade your work. This grading tool measures the following four categories: o Knowledge and Understanding: Measures how much you know and understand the content. Do you comprehend the meaning and significance of the content? o Inquiry and Thinking: Measures how you use information. Are you thinking about the question, planning your response and giving answers from many different perspectives? o Communication: Measures how you communicate your ideas for different audiences and purposes. Do you use relevant conventions, vocabulary and terminology? o Application: Measures your ability to apply what you have learned in this course to your own life and in other new situations. |
||
| Evaluation Table | ||
|
*Out of Score is the maximum points achievable in this evaluation item. *Weighted Max Score is the Out of Score converted to reflect its actual percentage weight in the course (8%). This ensures a fair grading system based on the assignment's importance in the overall course evaluation. |
||
| The Big Picture | ||
* Mid Term report cards will be released at this point of the course |
- 03 - Average Rates of Change.pdf
- 1.pdf
- 1310_Notes_5o4_fill2.PDF
- 2-3 Making comics.png
- 2-3 Symbolism preview pic.png
- 2-3 symbols.png
- 2.pdf
- 2017-05-17_0704.png
- 5-3DRa.png
- 5-3DRb.png
- 5-3DRc.png
- 5-3DRd.png
- 51.pdf
- 70-30-split.png
- 8-3_Practice_Answers.pdf
- Adding and Subtracting Functions (CW or HW).pdf
- composite_functions_intro.pdf
- Compound_Angles_WS.pdf
- courseoverview.png
- Dividing Polynomials (1).pdf
- Dividing Polynomials.pdf
- Download File (1).pdf
- Download File (2).pdf
- Download File.pdf
- Download File(1) (1).pdf
- Download File(1) (2).pdf
- Download File(1).pdf
- Download File(2).pdf
- Download File(3).pdf
- Download File(4).pdf
- Download File(5).pdf
- Download File(6).pdf
- ENG3U Course Outline 2018-19.pdf
- eng3u_kica.png
- eng3u-70-30-split.png
- eng3u-70-30.png
- Exercises_LogarithmicFunction.pdf
- files.jpg
- Function Operations.pdf
- Math0983-ExponentialAndLogarithmicEquations.pdf
- mc-bus-loglaws-2009-1.pdf
- module4.pdf
- notes.png
- Properties of Logarithms.pdf
- rational_functions_handout.pdf
- SCH4U Course Outline 2019-20.docx
- SCH4U Course outline 2022-2023.pdf
- SCH4U Course Outline.pdf
- Screen Shot 2020-01-13 at 10.02.33 AM.png
- Screen Shot 2020-07-31 at 1.02.36 PM.png
- Screen Shot 2020-11-27 at 4.38.37 PM.png
- Section 7.4 Reciprocals of Linear Functions (Part1).pdf
- SZ 6.5.pdf
- transformations_handout.pdf
- trigidentities1.pdf
- video.jpeg
- web-polynomialdivision.pdf
- ws log equations.pdf